| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 22:52:50 UTC |
|---|
| Update Date | 2020-04-22 15:10:15 UTC |
|---|
| BMDB ID | BMDB0002220 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
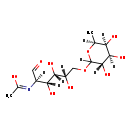
| Common Name | N-Acetyl-6-O-L-fucosyl-D-glucosamine |
|---|
| Description | N-Acetyl-6-O-L-fucosyl-D-glucosamine is an extremely weak basic (essentially neutral) compound (based on its pKa). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| N-[(2R,3R,4S,5S)-3,4,5-Trihydroxy-1-oxo-6-{[(3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}hexan-2-yl]ethanimidate | Generator |
|
|---|
| Chemical Formula | C14H25NO10 |
|---|
| Average Molecular Weight | 367.351 |
|---|
| Monoisotopic Molecular Weight | 367.147846009 |
|---|
| IUPAC Name | N-[(2R,3R,4S,5S)-3,4,5-trihydroxy-1-oxo-6-{[(3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}hexan-2-yl]ethanimidic acid |
|---|
| Traditional Name | N-[(2R,3R,4S,5S)-3,4,5-trihydroxy-1-oxo-6-{[(3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}hexan-2-yl]ethanimidic acid |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H][C@](O)(COC1([H])O[C@@]([H])(C)[C@@]([H])(O)[C@@]([H])(O)[C@]1([H])O)[C@@]([H])(O)[C@]([H])(O)[C@]([H])(C=O)N=C(C)O |
|---|
| InChI Identifier | InChI=1S/C14H25NO10/c1-5-9(19)12(22)13(23)14(25-5)24-4-8(18)11(21)10(20)7(3-16)15-6(2)17/h3,5,7-14,18-23H,4H2,1-2H3,(H,15,17)/t5-,7-,8-,9+,10+,11+,12+,13-,14?/m0/s1 |
|---|
| InChI Key | YBWAUUBLHOFOPK-RQKHFBPKSA-N |
|---|
| Chemical Taxonomy |
|---|
| Classification | Not classified |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | |
|---|