| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 22:48:50 UTC |
|---|
| Update Date | 2020-04-22 15:09:04 UTC |
|---|
| BMDB ID | BMDB0001902 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Pteroyltriglutamic acid |
|---|
| Description | Pteroyltriglutamic acid belongs to the class of organic compounds known as oligopeptides. These are organic compounds containing a sequence of between three and ten alpha-amino acids joined by peptide bonds. Based on a literature review a significant number of articles have been published on Pteroyltriglutamic acid. |
|---|
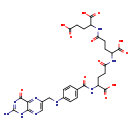
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Pteroyltriglutamate | Generator | | N-[N-(N-Pteroyl-L-gamma-glutamyl)-L-gamma-glutamyl]-glutamic acid | HMDB | | Folic acid diglutamate | HMDB | | N-[4-[[(2-amino-1,4-dihydro-4-oxo-6-Pteridinyl)methyl]amino]benzoyl]-L-gamma-glutamyl-L-gamma-glutamyl-L-glutamic acid | HMDB | | N-[N-[N-[4-[[(2-amino-1,4-dihydro-4-oxo-6-Pteridinyl)methyl]amino]benzoyl]-L-.gamma.-glutamyl]-L-.gamma.-glutamyl]- L-glutamic acid | HMDB | | Pteropterin | HMDB, MeSH | | Pteroyl-gamma-triglutamic acid | HMDB | | Pteroyl-L-glutamyl-gamma-L-glutamyl-gamma-L-glutamic acid | HMDB | | Pteroyltri-gamma-L-glutamic acid | HMDB | | Teropterin | MeSH, HMDB | | Pteropterin, sodium salt | MeSH, HMDB | | Pteroyl-gamma-glutamyl-gamma-glutamylglutamic acid | MeSH, HMDB | | 2-{[4-carboxy-4-({4-carboxy-1-hydroxy-4-[(4-{[(4-hydroxy-2-imino-1,2-dihydropteridin-6-yl)methyl]amino}phenyl)formamido]butylidene}amino)-1-hydroxybutylidene]amino}pentanedioate | Generator, HMDB | | Pteroyltriglutamic acid | MeSH |
|
|---|
| Chemical Formula | C29H33N9O12 |
|---|
| Average Molecular Weight | 699.6254 |
|---|
| Monoisotopic Molecular Weight | 699.224867565 |
|---|
| IUPAC Name | 2-(4-{4-[(4-{[(2-amino-4-oxo-1,4-dihydropteridin-6-yl)methyl]amino}phenyl)formamido]-4-carboxybutanamido}-4-carboxybutanamido)pentanedioic acid |
|---|
| Traditional Name | 2-(4-{4-[(4-{[(2-amino-4-oxo-1H-pteridin-6-yl)methyl]amino}phenyl)formamido]-4-carboxybutanamido}-4-carboxybutanamido)pentanedioic acid |
|---|
| CAS Registry Number | 89-38-3 |
|---|
| SMILES | NC1=NC(=O)C2=NC(CNC3=CC=C(C=C3)C(=O)NC(CCC(=O)NC(CCC(=O)NC(CCC(O)=O)C(O)=O)C(O)=O)C(O)=O)=CN=C2N1 |
|---|
| InChI Identifier | InChI=1S/C29H33N9O12/c30-29-37-23-22(25(44)38-29)33-15(12-32-23)11-31-14-3-1-13(2-4-14)24(43)36-18(28(49)50)6-9-20(40)34-16(26(45)46)5-8-19(39)35-17(27(47)48)7-10-21(41)42/h1-4,12,16-18,31H,5-11H2,(H,34,40)(H,35,39)(H,36,43)(H,41,42)(H,45,46)(H,47,48)(H,49,50)(H3,30,32,37,38,44) |
|---|
| InChI Key | WOLQREOUPKZMEX-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as oligopeptides. These are organic compounds containing a sequence of between three and ten alpha-amino acids joined by peptide bonds. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Oligopeptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-oligopeptide
- Gamma-glutamyl alpha-amino acid
- Glutamic acid or derivatives
- Glutamine or derivatives
- Tetracarboxylic acid or derivatives
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- Hippuric acid
- Hippuric acid or derivatives
- Pterin
- Alpha-amino acid or derivatives
- Pteridine
- Aminobenzamide
- Aminobenzoic acid or derivatives
- Benzoic acid or derivatives
- Benzamide
- Benzoyl
- Phenylalkylamine
- Aniline or substituted anilines
- Pyrimidone
- Aralkylamine
- Secondary aliphatic/aromatic amine
- Aminopyrimidine
- Monocyclic benzene moiety
- Fatty amide
- Fatty acyl
- N-acyl-amine
- Benzenoid
- Pyrazine
- Pyrimidine
- Vinylogous amide
- Heteroaromatic compound
- Secondary carboxylic acid amide
- Amino acid
- Amino acid or derivatives
- Carboxamide group
- Secondary amine
- Organoheterocyclic compound
- Azacycle
- Carboxylic acid
- Organic oxide
- Hydrocarbon derivative
- Organic nitrogen compound
- Organic oxygen compound
- Organonitrogen compound
- Organooxygen compound
- Primary amine
- Carbonyl group
- Organopnictogen compound
- Amine
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 0.1 mg/mL at 5 °C | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udi-0221229000-c23f869dc13b1d95272a | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_5) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_6) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_7) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_8) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_9) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_10) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_2) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_3) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_4) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_5) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_6) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_7) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_8) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_9) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_10) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_11) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_12) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_13) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_14) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0330009000-e987962787f2029e2f08 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0059-0960106000-21f9d9b3b6972db9bc7c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-0941002000-891cdeb849daa1e9d839 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0f8a-0000009000-bd7ecf2c2eeac6ea62e6 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0gws-0110019000-44f0bdd173f7125a9395 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f97-5941201000-3d500662a3c45848f410 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0290004200-8f2f396143788196bc73 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a5a-0771391000-85c9b3b383e3d558e48b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004j-0951010000-28b5c68dcb7492d5286e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-052s-0010009000-f025b4c029bb870e38f9 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0nni-0311109000-18e4bbbd67edf5591bc5 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0w4i-3924031000-85da69ca05ffb0871921 | View in MoNA |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | Not Available | View in JSpectraViewer |
|---|
|
|---|